g. throughout drug products improvement. It is the ideal fit for you In case the temperature, timepoints, or solvents encouraged because of the Sartorius method don't suit your goal. We build extractables information In keeping with your person demands.
By way of example: Product A has long been cleaned out. The products A has a standard each day dose of 10mg plus the batch measurement is 200 kg.
Suppliers should create a sampling prepare to find out the spots and frequency of sampling throughout the cleaning method. The system ought to take into consideration the two Visible inspection and analytical tests to make certain that all residues and contaminants are effectively removed.
— breakdown items in the detergents, acids and alkalis That could be applied as Portion of the cleaning method.
Anytime introduction, elimination or modification of any tools evaluation /assessment shall be finished According to annexure no. II, or
Each individual is a top quality-managed and authorized doc which can be used to qualify design and set up qualification and to ascertain the need for leachables tests.
Cleaning validation: A long-time period research to determine that cleaning processes consistently produce appropriate final results.
Stainless steel/ Teflon/ Silicon/ PVC etcetera templates shall be employed for determining the surface area region from the swab, or eyeball method be practiced and validated for each sampling personals
Q. What would you imply by pre-formulation review? A analyze conducted prior to the formulation development starts and aim to collect Principal specifics of the Actual physical and chemical Homes of the drug material, drug product or service and reference product or service (RLD). Q. What does one imply by RLD? RLD stands for Reference Mentioned Drug accredited by regulatory authority…
A cleaning validation for any loved ones of compounds utilizing swab sampling and rinse Answer methods, and large overall performance liquid chromatography for separation and detection from the analytes was carried out.Successful parameters on recovery which includes sampling method, swab features, solvent, swabbing strategy, and material substance of products Call surfaces inside the production devices for swab and rinse sampling method, quantitative cleaning verification method, and active pharmaceutical component (API) amount and mother nature happen to be researched.The limit of detection along with the limit of quantitation to the HPLC method were identified for being 0.0198 µg/mL, and 0.0495 µg/mL of your analyte, respectively.
Clinical pharmacists play a vital purpose in healthcare settings, they are specialised in the choice of medicines and making sure their Protected and successful use.
Immersion Method: The immersion method might be both agitated, the place a cleaning agent inside a approach vessel is mechanically stimulated, or static, wherever the method vessel is soaked With all get more info the cleaning agent.
All corporations & industries need unique frequencies for cleaning validation. Production products complexity & the type of merchandise manufactured affect frequency.
With an Extractables Evaluation, substances located in the typical extractables analyses under acceptable worst-circumstance conditions can be evaluated for person shopper processes. This supplies you an extensive comprehension of, and self-assurance in, the Sartorius parts check here utilised with your processes.
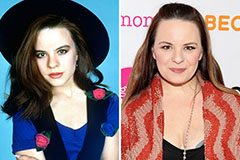 Jenna Von Oy Then & Now!
Jenna Von Oy Then & Now! Freddie Prinze Jr. Then & Now!
Freddie Prinze Jr. Then & Now! Michelle Trachtenberg Then & Now!
Michelle Trachtenberg Then & Now! Elisabeth Shue Then & Now!
Elisabeth Shue Then & Now! Earvin Johnson III Then & Now!
Earvin Johnson III Then & Now!